DLCO is one of the most useful numbers in lung testing — but it’s also one of the most misunderstood. Many patients and even clinicians notice DLCO values “bounce” between tests. Is that real change or just measurement noise?.
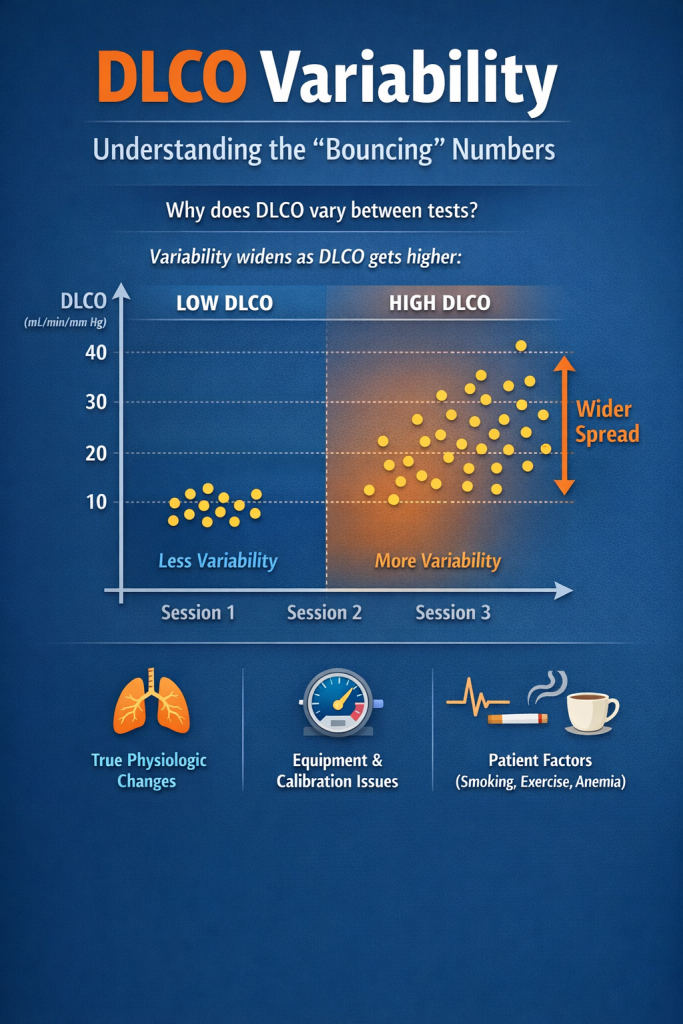
Recent evidence shows that inter-session differences can exceed traditional thresholds for a meaningful change. Absolute shifts beyond 3.5 mL/min/mm Hg, 15 percent of predicted, or 21 percent relative are not rare, especially at higher DLCO values where variability widens. Variability is not constant. At higher DLCO values the amount of scatter between tests tends to increase.. That reality calls for a more nuanced, baseline-aware framework grounded in your laboratory’s own reproducibility.
This guide explains what DLCO measures, why it varies, how to build ATS/ERS-aligned quality systems, and how to interpret change using baseline-specific, evidence-linked limits. We finish with practical tips on when to repeat testing and how to integrate imaging and symptoms for decisions that matter.
What DLCO measures and why it matters
DLCO (diffusing capacity of the lung for carbon monoxide) quantifies the transfer of carbon monoxide from alveolar gas into pulmonary capillary blood during a brief breath-hold maneuver. It reflects the combined effects of:
- Alveolar-capillary membrane conductance
- Pulmonary capillary blood volume
- Hemoglobin (Hb) concentration and binding
- Ventilation-perfusion matching
Because many lung disorders alter one or more of these components, DLCO helps differentiate emphysema from asthma, identify interstitial lung disease earlier than spirometry, and monitor disease activity and treatment effects. DLCO is typically interpreted alongside alveolar volume (VA), the DLCO/VA ratio (often reported as KCO), and spirometry.
Why DLCO varies between sessions
Variability arises from both technical and biological sources, and it grows with higher DLCO values.
Technical contributors include:
- Breath-hold timing: Small deviations from the recommended 10-second breath hold alter diffusion time.
- Inspired volume: Suboptimal inspiratory vital capacity lowers VA and alters DLCO and KCO.
- Analyzer calibration and leak integrity: Gas analyzers and sample pathways require tight calibration; small leaks distort tracer gas fractions.
- Ambient conditions: Barometric pressure and temperature corrections must be accurate.
- Hemoglobin correction: DLCO should be adjusted for current Hb; using an old value can mislead.
- Equipment drift: Even well-maintained systems exhibit signal drift that must be tracked and corrected.
Biological contributors include:
- Day-to-day pulmonary vascular tone and cardiac output
- Airway tone and small changes in bronchodilation
- Posture, recent exertion, or caffeine
- Anemia, dehydration, or fluid shifts
- Smoking on the day of test (elevates carboxyhemoglobin and can reduce DLCO)
The practical upshot: inter-session differences frequently exceed the traditional absolute (3.5 mL/min/mm Hg) or relative (15 percent predicted, 21 percent relative) cutoffs, particularly when baseline DLCO is high. That heteroscedasticity argues for interpretation anchored to your lab’s precision profile.
Building reliability: ATS/ERS-aligned quality assurance
High-quality DLCO starts with consistent people, procedures, and proof that the system is behaving. Key steps that align with American Thoracic Society (ATS) and European Respiratory Society (ERS) guidance include:
- Daily 3-L syringe checks: Verify volume accuracy and leak integrity for flow sensors and the DLCO system.
- Weekly DLCO syringe quality control (QC): Use certified gas mixtures and a DLCO simulator or syringe method to confirm analyzer response and timing logic.
- Monthly biologic controls: Test a healthy, stable staff member or designated control subject to track real-world reproducibility over time.
- Calibration and environmental logging: Document barometric pressure, temperature, and gas analyzer calibrations before patient testing.
- Suspension on QC failure: If any QC step fails, stop patient testing, troubleshoot, and document corrective action before resuming.
- Standardized coaching: Use a consistent script for pre-breathing, rapid inspiration to total lung capacity, breath-hold timing, and relaxed complete exhalation.
These steps do more than create neat logs. They generate the data you need to calculate your own reproducibility limits and to defend clinical interpretations.
Interpreting change: a baseline-specific framework
Given heteroscedasticity, a single universal cutoff can either undercall change at low baselines or overcall it at high baselines. A pragmatic approach is to classify change relative to both the patient’s baseline and your lab’s measured variability.
Recommended structure:
- Stable: Change falls within your lab’s reference change value (RCV) at the patient’s baseline range.
- Possible change: Exceeds the RCV but falls within expanded limits suggested by multi-visit evidence for that baseline band.
- Definite change: Surpasses both the RCV and evidence-linked expanded limits, or is corroborated by parallel shifts in VA, KCO, symptoms, and imaging.
How to set thresholds:
- Quantify lab reproducibility. From monthly biologic controls and repeat patient testing, compute coefficient of variation (CV) across DLCO strata. Expect higher CV at higher DLCO.
- Derive RCV. For each stratum, RCV ≈ 1.96 × √2 × CV. This gives the minimum change likely to exceed measurement noise with 95 percent confidence.
- Anchor to evidence. Compare your stratum-specific RCVs to published limits around 3.5 mL/min/mm Hg absolute, 15 percent predicted, and 21 percent relative. Allow wider limits where your data and the literature show heteroscedastic spread.
- Apply clinical weighting. Favor Definite change classification when multiple signals agree: DLCO and KCO fall together, VA is stable, symptoms progress, and imaging aligns.
This framework respects local precision while remaining consistent with broader evidence.
Practical tips for clinicians and labs
- Standardize timing. Repeat testing at similar times of day, similar posture, and with consistent pre-test instructions about caffeine, exertion, and smoking abstinence.
- Check hemoglobin. Reconfirm Hb when clinical context suggests change; apply the appropriate correction consistently.
- Integrate context. Pair DLCO trends with symptoms, high-resolution computed tomography (HRCT), echocardiography when pulmonary hypertension is suspected, and therapy changes.
- Repeat when uncertain. If an isolated DLCO shift lands in Possible change, repeat within 2 to 8 weeks, ideally with the same technologist and equipment, to confirm direction and magnitude.
- Document acceptability. Ensure at least two acceptable bites with inspiratory volume near vital capacity and repeatability within the lab’s predefined limits.
- Over-reading matters. Systematic over-reading by an experienced interpreter links QC findings with final clinical statements, explicitly noting when measurement precision constrains conclusions.
How our lab approach translates to actionable results
Understanding DLCO variability is especially important when testing is used to guide training or health decisions outside a hospital setting. At Metatec, respiratory-therapy-led staff emphasize rigorous QC and clear interpretation across all gas-exchange testing we perform. For athletes and health-focused adults, that means you leave with data you can trust and practical next steps for training, recovery, and nutrition. If you are exploring aerobic capacity or thresholds, learn more about our options for oxygen uptake testing that includes detailed zone guidance by visiting our page on oxygen uptake testing. If nutrition and metabolism are your focus, see how an indirect calorimetry session can clarify calorie targets by visiting our page on metabolic testing.
FAQ: quick answers to common questions
- What is DLCO?
DLCO (diffusing capacity of the lung for carbon monoxide) measures how effectively gas moves from the alveoli into the blood during a brief breath hold. It reflects membrane conductance, capillary blood volume, hemoglobin, and ventilation-perfusion matching. - What drives variability between DLCO sessions?
Both technical factors (breath-hold timing, inspired volume, analyzer calibration, ambient pressure, Hb correction, equipment leaks or drift) and biological factors (cardiac output, airway tone, smoking, recent exercise, anemia or hydration status) contribute. Variability typically increases as DLCO gets higher. - How should clinicians interpret meaningful change in DLCO?
Use a baseline-specific approach anchored to your lab’s RCV, then classify results as Stable, Possible change, or Definite change. Consider absolute and relative differences, heteroscedastic patterns, and corroboration from VA, KCO, symptoms, and imaging. - What quality control practices improve DLCO reliability?
Daily 3-L syringe checks, weekly DLCO syringe QC, monthly biologic controls, strict calibration and environmental logging, standardized coaching, and suspension of testing on QC failure. - How much does it cost to get your metabolism tested?
Pricing can vary by location, equipment, and whether you choose a package. Our site features periodic promotions rather than a permanent list. For current options, see our page that covers calorie needs testing. - Is the RMR test covered by insurance?
Resting metabolic rate (RMR) testing is typically a self-pay service and is often not covered by health insurance. Coverage policies vary, so check with your insurer directly. - Does insurance cover metabolic testing?
In most fitness and wellness contexts, metabolic testing is not covered and is paid out of pocket. Medical indications ordered by a clinician may be handled differently by some plans; confirm with your provider and insurer. - What are the symptoms of slow metabolism?
Low resting energy expenditure can present as fatigue, difficulty losing weight despite consistent effort, feeling cold, and low exercise tolerance. These symptoms are non-specific and can result from many causes. Objective testing with indirect calorimetry helps differentiate true low resting metabolic rate from other factors.
When to repeat testing and how to act on results
- Repeat promptly if a DLCO shift lands in Possible change or if test quality flags appear on review.
- Repeat after clinical inflection points, such as treatment initiation or a major symptom change.
- Reassessing when Hb changes or when imaging suggests a new process that could alter diffusion.
Integrate the repeat measurement with symptoms, imaging, and parallel physiologic data. A confirmed Definite change warrants revisiting the differential diagnosis, therapy, or further testing.
Summary and next steps
DLCO is indispensable, but it is not noise-free. Real-world data show that variability widens as DLCO increases, which can push changes beyond traditional cutoffs. The answer is not to ignore movement, but to ground interpretation in ATS/ERS-aligned quality systems, compute laboratory-specific RCVs, and classify change relative to baseline and corroborating evidence. That approach protects patients from overcalling noise and helps clinicians act confidently when change is real.
If you are aligning gas-exchange insights with training or nutrition decisions, our team pairs rigorous QC with clear, actionable reporting.
